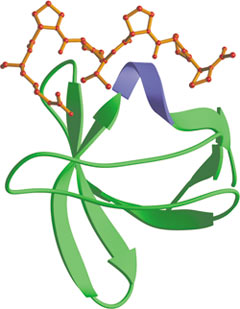
The C-terminal SH3 domain of SEM-5 bound to mSOS peptide (red).
Src-homology 3 (SH3) domains bind to Pro-rich peptides that form a left-handed poly-Pro type II helix, with the minimal consensus Pro-X-X-Pro. An aliphatic residue usually precedes each proline, with each of the aliphatic-Pro pairs binding to a hydrophobic pocket on the SH3 domain. In principle, the ligand can bind in either orientation. An additional non-Pro residue (frequently Arg) contacts the SH3 domain and can form part of the binding core. Such peptides usually bind to the SH3 domain with a Kd in the µM range. The binding affinity and specificity can be markedly increased by tertiary interactions involving loops on the SH3 domain.
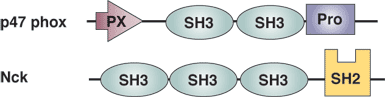
| SH3 Domain Proteins | Binding Partners | SH3 Domain Binding Sites |
| Src Tyrosine Kinase | p85 subunit of Pl3 kinase | RPLPVAP Class l N-terminal to C-terminal binding site |
| Crk Adaptor Protein | C3G guanidine nucleotide exchanger | PPPALPPKKR Class ll C-terminal to N-terminal binding site |