CAR-Engineered Cell Characterization Solutions
The development of chimeric antigen receptor (CAR) -based cell therapies is revolutionizing the treatment of human cancers and autoimmune diseases. As this innovative therapeutic modality continues to improve and become more accessible, better reagents are improving our ability to detect, identify, and fully characterize CAR-expressing cells. With a growing portfolio of innovative products, CST is helping to advance researchers' work in this field.
Characterize Your CAR-Engineered Cells Easier and Faster
One of the most significant challenges facing scientists developing and characterizing CAR-engineered cells is the need for robust, sensitive, and selective research reagents for the identification of CAR-engineered cells. If you plan to use an immunoassay platform in your research, developing your own anti-idiotype antibodies to identify CAR-engineered cells can be a long and resource-intensive process.
A Workflow Solution for Characterization of CAR-Engineered Cells
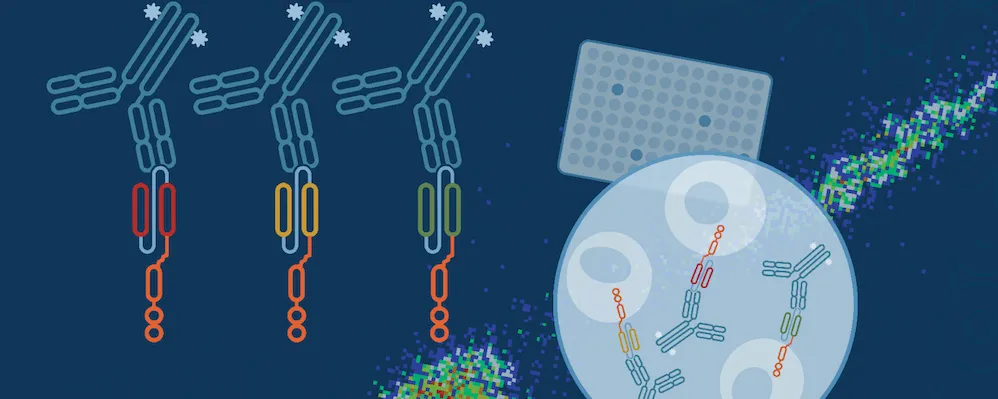
There are many stages in the complex process of characterizing CAR-engineered cells. CST focuses on one critical step in the process with a suite of purpose-built research tools for CAR cell characterization.
Your streamlined CAR-engineered cell characterization workflow starts here →
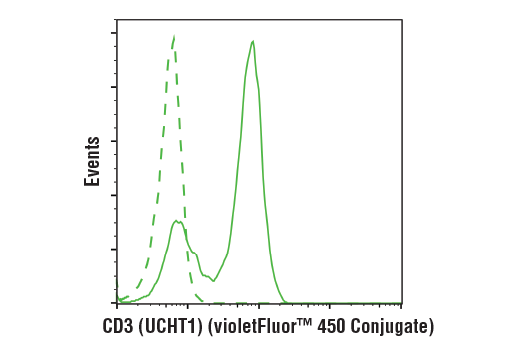
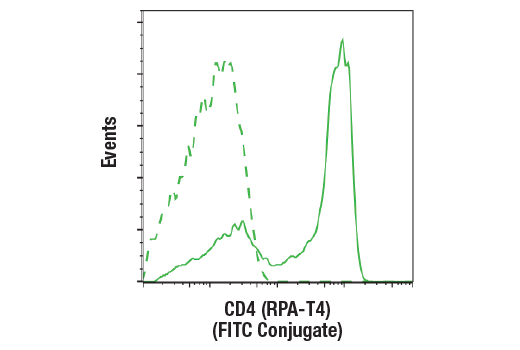
DETECT
Quickly and easily evaluate CAR and target antigen expression
ANALYZE
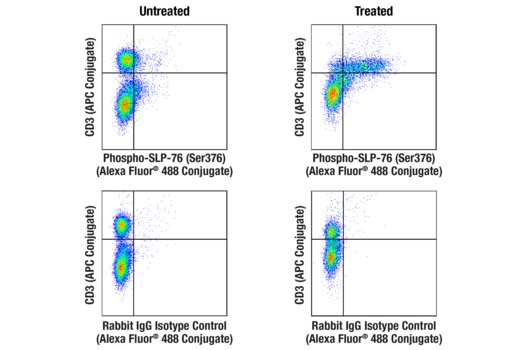
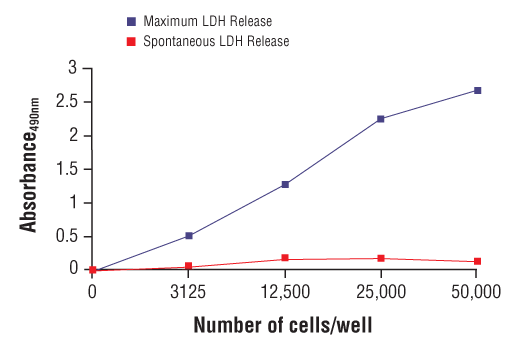
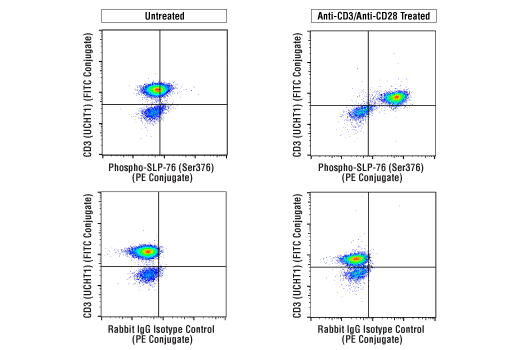
Simplify immune cell activation, proliferation, viability, and signaling assays
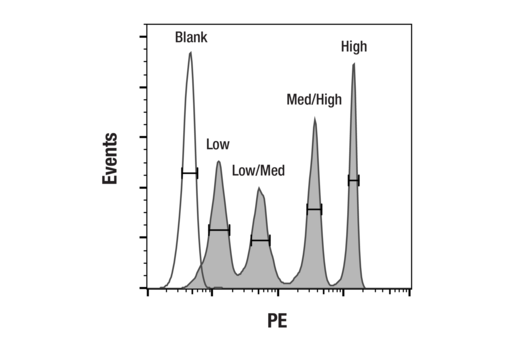
QUANTITATE
Streamline measurement of CAR transduction efficiency and cell surface expression
PURIFY
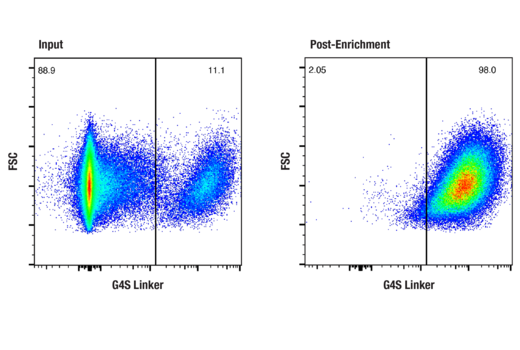
Enrich CAR+ cells using either bead-based or FACS-based sorting with high specificity
TRANSLATE
Directly interrogate the tumor microenvironment to measure infiltration of CAR-engineered cells
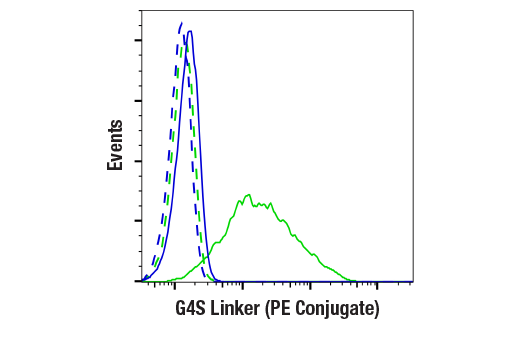
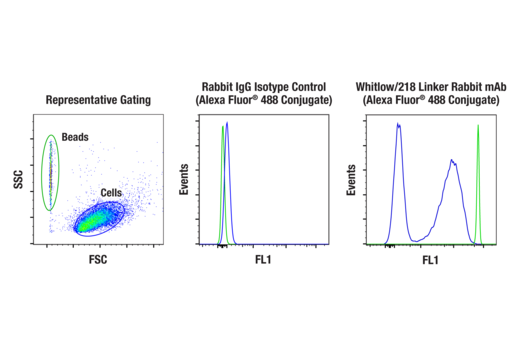
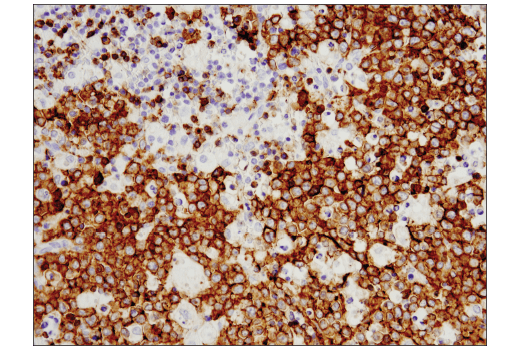
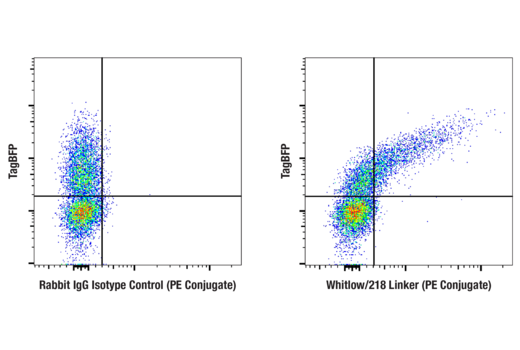
To streamline your characterization assays, CST scientists developed recombinant monoclonal antibodies to the ubiquitous peptide linker sequences of scFv-based CARs. By targeting these ubiquitous linker sequences, you can significantly reduce time, effort, and cost by eliminating the need for anti-idiotype antibodies to every CAR variant you’re testing. With high specificity to either the G4S or Whitlow/218 linkers, our CAR linker antibodies can detect the expression of scFv-based CARs, regardless of scFv specificity.
Video: Detecting CAR Expression with Versatile, Innovative Antibodies (2:04)
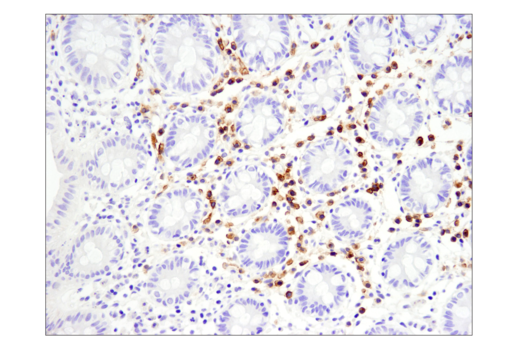
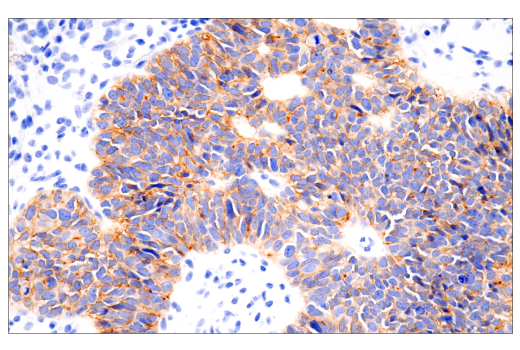
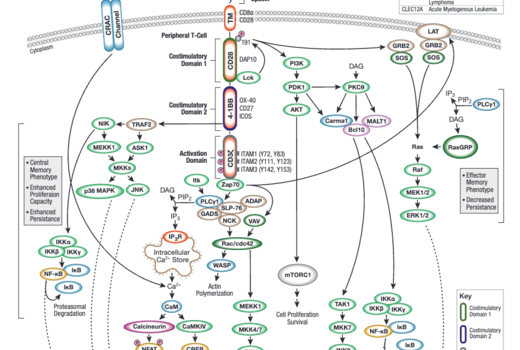
Tools for Translational Research
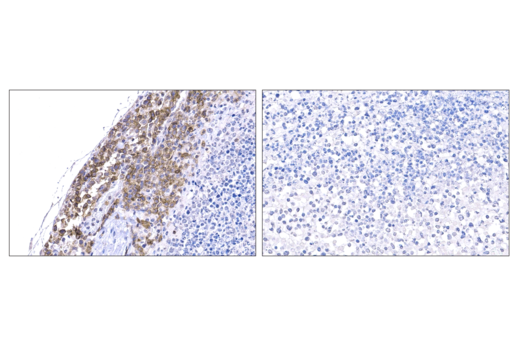
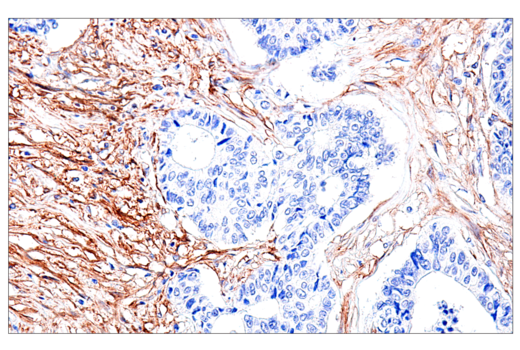
When challenging solid tumors with CAR-engineered cells, a critical question is whether these cells are capable of entering the tumor microenvironment. Traditional methods have significant drawbacks. It is fortunate that direct interrogation of CAR protein expression in solid tumors using commercially available antibodies is now possible.
CAR-Engineered Cell Characterization Resources
Explore a growing library of Webinars, Scientific Posters, Application Notes, and other resources focused on CAR cell characterization.