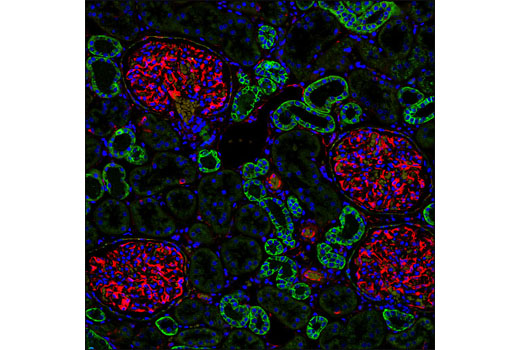
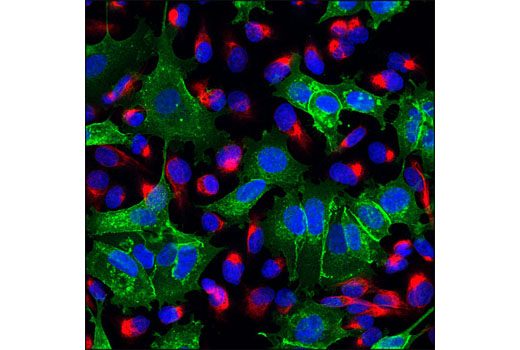
| Products Included | No. | Volume | Applicaton | Dilution | Reactivity | Homology† |
|---|---|---|---|---|---|---|
| PathScan® EMT Duplex IF Kit Primary Antibody Cocktail | 7783 | 100 µl | Immunofluorescence (Immunocytochemistry), Immunofluorescence (Paraffin) | 1:100 | Human | Mouse |
| PathScan® Duplex IF Kit Detection Cocktail I | 7832 | 100 µl | Immunofluorescence (Immunocytochemistry), Immunofluorescence (Paraffin) | 1:100 | N/A | N/A |
†Species predicted to react based on 100% sequence homology.
| Kit Analytes | Detection Dye | Ex(max) (nm) | Em(max) (nm) |
|---|---|---|---|
| E-cadherin | Alexa Fluor® 488 | 495 | 519 |
| Vimentin | Alexa Fluor® 555 | 555 | 565 |
IF-P, IF-IC
#P08670, #P12830
7431, 999
Product Information
Storage
Specificity / Sensitivity
Species Reactivity:
Human
Source / Purification
Monoclonal antibodies were produced by immunizing animals with a synthetic peptide corresponding to residues surrounding Arg45 of human vimentin protein or residues surrounding Pro780 of human E-cadherin protein.
Product Description
Background
Epithelial-mesenchymal transition (EMT) refers to a biological process in which cells undergo a series of biochemical changes that induce a morphological transformation from an epithelial, polarized, adhesive state to an irregular, elongated, mesenchymal phenotype that enables migratory capacity (1,2). EMTs are classified into three subtypes: those involved in implantation, embryogenesis, and organ development; those associated with inflammation and fibrosis; and those involved in invasion and metastasis (1). Molecular changes that are associated with cells during this transformation include the loss of E-cadherin and gain of vimentin expression, hallmark epithelial and mesenchymal markers, respectively (3-5). Numerous studies have established that EMT is an essential step in cancer metastasis (5-7). E-cadherin is regarded as an active suppressor of invasion and tumorigenesis (8). In response to extracellular stimuli, vimentin coordinates various signaling pathways to induce spatial reorganization and structural changes (9), reminiscent of the EMT phenotype observed in motile cells involved in invasion and metastasis (6).
- Kalluri, R. and Weinberg, R.A. (2009) J Clin Invest 119, 1420-8.
- Lee, J.M. et al. (2006) J Cell Biol 172, 973-81.
- Yan, W. et al. (2010) J Biol Chem 285, 14042-51.
- Sethi, S. et al. (2011) Transl Oncol 4, 222-6.
- Kim, J.H. et al. (2007) J Korean Med Sci 22, 898-904.
- Emadi Baygi, M. et al. (2010) Cell Biol Toxicol 26, 553-67.
- Sethi, S. et al. (2010) Am J Transl Res 3, 90-9.
- Wheelock, M.J. and Johnson, K.R. (2003) Annu Rev Cell Dev Biol 19, 207-35.
- Helfand, B.T. et al. (2004) J Cell Sci 117, 133-41.
Species Reactivity
Species reactivity is determined by testing in at least one approved application (e.g., western blot).
Applications Key
IF-P: Immunofluorescence (Paraffin) IF-IC: Immunofluorescence (Immunocytochemistry)
Cross-Reactivity Key
H: human M: mouse R: rat Hm: hamster Mk: monkey Vir: virus Mi: mink C: chicken Dm: D. melanogaster X: Xenopus Z: zebrafish B: bovine Dg: dog Pg: pig Sc: S. cerevisiae Ce: C. elegans Hr: horse GP: Guinea Pig Rab: rabbit All: all species expected
Trademarks and Patents
Limited Uses
Except as otherwise expressly agreed in a writing signed by a legally authorized representative of CST, the following terms apply to Products provided by CST, its affiliates or its distributors. Any Customer's terms and conditions that are in addition to, or different from, those contained herein, unless separately accepted in writing by a legally authorized representative of CST, are rejected and are of no force or effect.
Products are labeled with For Research Use Only or a similar labeling statement and have not been approved, cleared, or licensed by the FDA or other regulatory foreign or domestic entity, for any purpose. Customer shall not use any Product for any diagnostic or therapeutic purpose, or otherwise in any manner that conflicts with its labeling statement. Products sold or licensed by CST are provided for Customer as the end-user and solely for research and development uses. Any use of Product for diagnostic, prophylactic or therapeutic purposes, or any purchase of Product for resale (alone or as a component) or other commercial purpose, requires a separate license from CST. Customer shall (a) not sell, license, loan, donate or otherwise transfer or make available any Product to any third party, whether alone or in combination with other materials, or use the Products to manufacture any commercial products, (b) not copy, modify, reverse engineer, decompile, disassemble or otherwise attempt to discover the underlying structure or technology of the Products, or use the Products for the purpose of developing any products or services that would compete with CST products or services, (c) not alter or remove from the Products any trademarks, trade names, logos, patent or copyright notices or markings, (d) use the Products solely in accordance with CST Product Terms of Sale and any applicable documentation, and (e) comply with any license, terms of service or similar agreement with respect to any third party products or services used by Customer in connection with the Products.