Revision 1
#23682
Store at -20C
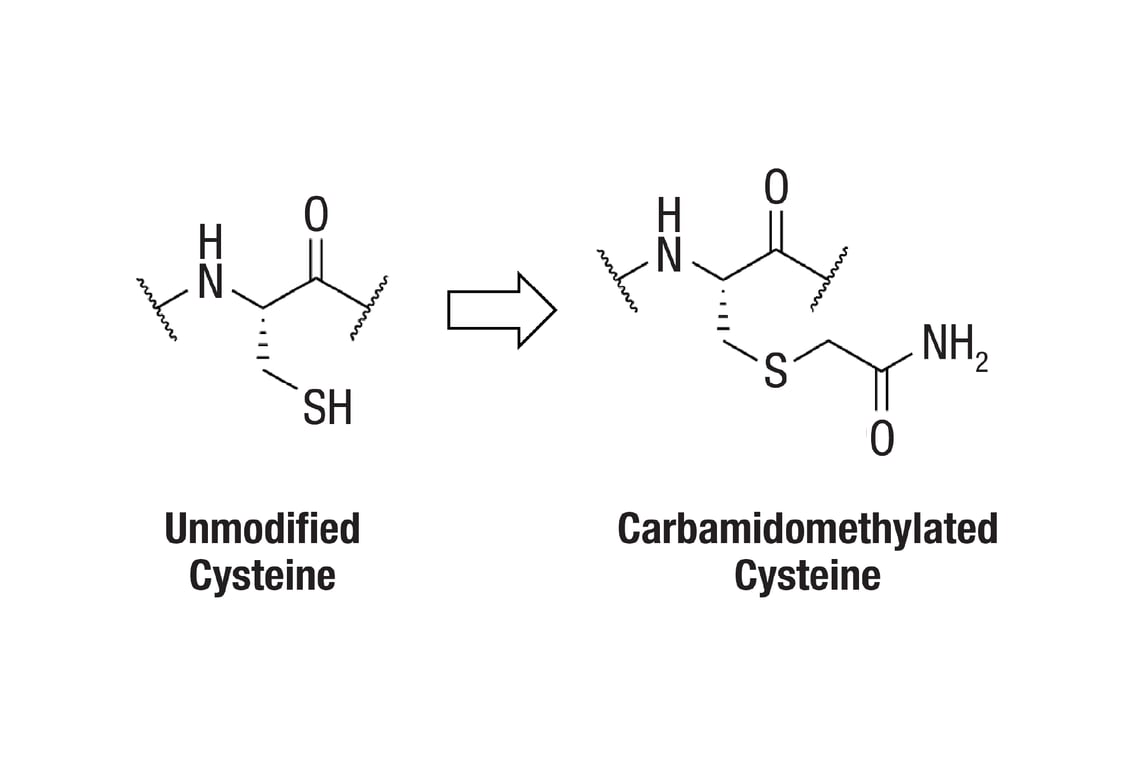
PTMScan® Carbamidomethyl Cysteine Motif (CAM-C) Kit
1 Kit
(10 assays)
877-616-CELL (2355)
877-678-TECH (8324)
3 Trask Lane | Danvers | Massachusetts | 01923 | USA
For Research Use Only. Not for Use in Diagnostic Procedures.
| Product Includes | Product # | Quantity | Storage Temp |
|---|---|---|---|
| PTMScan® IAP Buffer (10X) | 9993 | 10 x 600 µl | -20C |
| PTMScan® Carbamidomethyl Cysteine Motif (CAM-C) Immunoaffinity Beads | 90843 | 10 x 80 µl | -20C |
Description
Storage
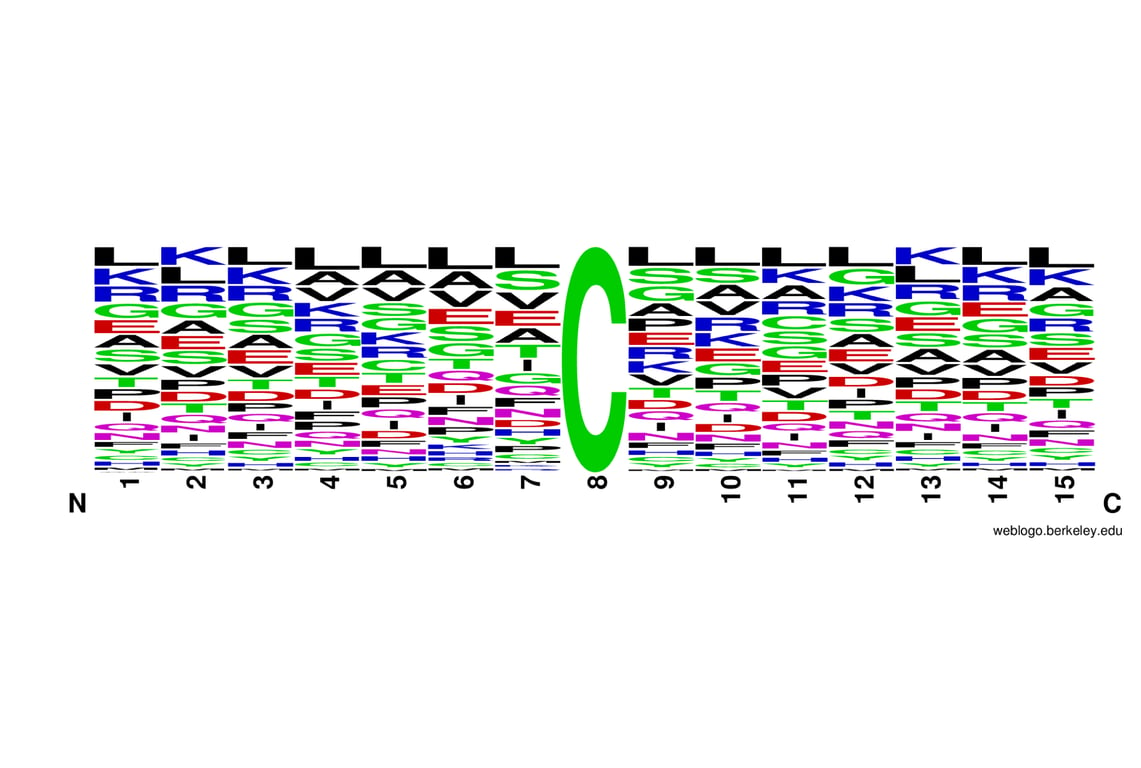
Background
Proteins are often derivatized to prevent disulfide bond scrambling, which can lead to artifactual bands in gel electrophoresis, and to minimize protease inaccessibility of cysteine-containing regions, which can limit coverage in mass spectrometry-based proteomics. In light of various studies describing potential non-cysteine reactivities of IAA and CAA and re-evaluating the effectiveness of such reactions, users should consider the benefits of cysteine derivatizations in relation to their specific experimental goals (2-4).
Trademarks and Patents
Cell Signaling Technology is a trademark of Cell Signaling Technology, Inc.
PTMScan is a registered trademark of Cell Signaling Technology, Inc.
All other trademarks are the property of their respective owners. Visit cellsignal.com/trademarks for more information.
Limited Uses
Except as otherwise expressly agreed in a writing signed by a legally authorized representative of CST, the following terms apply to Products provided by CST, its affiliates or its distributors. Any Customer's terms and conditions that are in addition to, or different from, those contained herein, unless separately accepted in writing by a legally authorized representative of CST, are rejected and are of no force or effect.
Products are labeled with For Research Use Only or a similar labeling statement and have not been approved, cleared, or licensed by the FDA or other regulatory foreign or domestic entity, for any purpose. Customer shall not use any Product for any diagnostic or therapeutic purpose, or otherwise in any manner that conflicts with its labeling statement. Products sold or licensed by CST are provided for Customer as the end-user and solely for research and development uses. Any use of Product for diagnostic, prophylactic or therapeutic purposes, or any purchase of Product for resale (alone or as a component) or other commercial purpose, requires a separate license from CST. Customer shall (a) not sell, license, loan, donate or otherwise transfer or make available any Product to any third party, whether alone or in combination with other materials, or use the Products to manufacture any commercial products, (b) not copy, modify, reverse engineer, decompile, disassemble or otherwise attempt to discover the underlying structure or technology of the Products, or use the Products for the purpose of developing any products or services that would compete with CST products or services, (c) not alter or remove from the Products any trademarks, trade names, logos, patent or copyright notices or markings, (d) use the Products solely in accordance with CST Product Terms of Sale and any applicable documentation, and (e) comply with any license, terms of service or similar agreement with respect to any third party products or services used by Customer in connection with the Products.
Revision 1
Revision 1
PTMScan Trypsin Digestion (small scale)
A. Solutions and Reagents
Reagents Not Included:
- 200 mM HEPES, pH 8.0 #44686
- Phosphatase Inhibitor Cocktail (100X) #5870
- Urea, Ultrapure, PTMScan® Qualified #60055
- Iodoacetamide, PTMScan® Qualified #88931
- DTT (Dithiothreitol) #7016
- PTMScan® Trypsin, TPCK-Treated #56296
- MS-Grade Trifluoroacetic Acid (TFA)
- LC-MS-Grade Acetonitrile (ACN)
- Sep-Pak Vac 1cc (50 mg) C18 Cartridge (Waters #WAT054955)
- Water, LC-MS Grade (Burdick and Jackson) #27732
- Phosphate Buffered Saline (PBS-20X) #9808
- 1 mM Hydrochloric Acid (HCl)
- Ammonium Bicarbonate, 1M in HPLC-Grade Water #78450
- BCA Protein Assay Kit #7780
- Peptide Desalting C18 StageTip #41745
- End-over-end Rotator
- Vacuum Concentrator (e.g., Speed-Vac)
NOTE: Prepare solutions using reverse osmosis deionized (RODI) or equivalent grade water for cell lysis (Section I). For Sections II-IV, prepare solutions using MS-grade water #27732.
Stock Solutions:
- Dithiothreitol (DTT): Make 1.25 M stock: 192.8 mg/1 mL, then divide into 20 µL aliquots. Store at -20°C for up to one year. Thaw one aliquot for each experiment.
- Trypsin-TPCK: Store dry powder for up to two years at -80°C. Seal the cap of the trypsin-TPCK container with parafilm to avoid collecting moisture, which can lead to degradation of the reagent. Prepare 1 mg/mL stock in 1 mM HCl. Divide into 1 mL aliquots. Store at -80°C for up to one year.
I. Cell Lysis and Protein Digestion
PTMScan® is compatible with many protein extraction, digestion, and purification protocols. Regardless of the particular method selected, ensure that peptides are completely dry and free of lysis buffer components, lipids, and excess salts prior to using the immunoaffinity purification kit. Below is a general protocol that uses in-solution digestion followed by solid phase extraction on Sep-Paks. However, due to the amount of input peptide needed and the type of labeling performed, the steps below can be adapted or scaled appropriately.
A. Solutions and Reagents
- Urea Lysis Buffer: 9 M urea, 20 mM HEPES (pH 8.0), 2X Phosphatase Inhibitor Cocktail. For 10 mL, weigh out 5.41 g of urea powder and dissolve it in 1 mL of 200 mM HEPES (pH 8.0) stock and 4 mL RODI water. Mix well until the urea goes into solution completely. Add 200 µL of Phosphatase Inhibitor Cocktail (100X), and adjust the final Urea Lysis Buffer volume to 10 mL total, such that the final concentration of phosphatase inhibitors is 2X.
- DTT solution (1.25 M): See the "Stock Solutions" section above for instructions on preparation.
- Iodoacetamide (IAA) solution (0.2 M): Dissolve 19 mg of IAA in water to a final volume of 0.5 mL. After weighing the powder, store in the dark and add water only immediately before use.
NOTE: The Urea Lysis Buffer should be prepared fresh prior to each experiment. Do not include protease inhibitors.
NOTE: Dissolving urea is an endothermic reaction. Urea Lysis Buffer preparation can be facilitated by placing a stir bar in the beaker and using a warm (not hot) water bath on a stir plate. 9 M urea is used so that upon lysis, the final concentration is approximately 8 M. The Urea Lysis Buffer should be used at room temperature.
IMPORTANT: Do not place the Urea Lysis Buffer on ice, as urea would precipitate out of solution.
NOTE: The IAA solution should be prepared fresh prior to each experiment.
B. Preparation of Suspension or Adherent Cell Lysate
- Grow approximately 1 x 106 cells for each experimental condition or enough cells to produce approximately 0.1 mg of soluble protein. (Note that 0.01 mg of soluble protein can be sufficient for carbamidomethyl cysteine enrichment.)
- Harvest suspension cells by centrifugation at 130 x g for 5 min at room temperature. Carefully remove supernatant, wash cells with cold 1X PBS, centrifuge, remove PBS wash, and add enough Urea Lysis Buffer (room temperature) to submerge the cell pellet, for instance, 0.1 mL of Urea Lysis Buffer. Pipette the slurry up and down a few times.
- Harvest adherent cells by decanting media, rinse with cold 1X PBS, and add enough Urea Lysis Buffer (room temperature) to submerge the plate surface, for instance, 0.1 mL of Urea Lysis Buffer. Pipette the slurry up and down a few times
IMPORTANT: Do not cool lysate on ice, as urea would precipitate out of solution.
IMPORTANT: Do not cool lysate on ice, as urea would precipitate out of solution.
C. Preparation of Tissue Lysate
- Collect tissue specimens by flash freezing in liquid nitrogen. A specimen of approximately 2-5 mg (wet weight) should provide sufficient material for Carbamidomethyl Cysteine enrichment (10 µg of soluble protein or more).
- Place the still-frozen tissue in a microcentrifuge tube. Add enough Urea Lysis Buffer to submerge it completely.
- Lyse the tissue using a homogenizer. Wash the tool with RODI water in between each sample
IMPORTANT: Do not cool lysate on ice, as urea would precipitate out of solution.
NOTE: If desired, the PTMScan® protocol may be interrupted at this stage. The cell lysate can be frozen and stored at -80°C for several weeks.
D. Sonication and Centrifugation
- Using a microtip, sonicate lysate at up to 15 W output with up to three bursts of 15 sec each. Cool on ice briefly between each burst.
- Clear the lysate by centrifugation at 4,000 - 10,000 x g for 15 min at room temperature and transfer the protein extract (supernatant) into a new microcentrifuge tube.
E. Protein Quantitation
- Measure soluble protein concentration using a colorimetric assay kit that is compatible with Urea Lysis Buffer. The BCA assay is recommended.
- Normalize all samples to prepare equal amounts of protein for each condition. Add Urea Lysis Buffer to increase the volume of all samples to the same amount. In the steps below, we illustrate with an example of 278 µL final volume.
F. Protein Reduction and Alkylation
- Dilute 1.25 M DTT two-fold with RODI water for 0.61 M DTT. Add 1/139 volume of 0.61 M DTT to the cleared lysate to reach a final concentration of 4.4 mM DTT (e.g., 2 µL of the diluted 0.61 M DTT to 278 µL lysate for a final volume of 280 µL). Mix well and incubate at room temperature for 60 min.
- Add 1/20 volume of 0.2 M IAA solution to the cleared lysate to reach a final concentration of 9 mM IAA (e.g., 14 µL of 0.2 M IAA to 280 µL lysate for a final volume of 294 µL). Mix well and incubate for 15 min at room temperature in the dark.
G. Protease Digestion
- Dilute 200 mM HEPES (pH 8.0) stock ten-fold with water to a final concentration of 20 mM HEPES (pH 8.0).
- Dilute reduced and alkylated lysate 4-fold with 20 mM HEPES (pH 8.0) to a final concentration of approximately 2 M urea, 20 mM HEPES (pH 8.0). For example, add 882 µL of 20 mM HEPES (pH 8.0) to 294 µL of lysate for a final volume of approximately 1.2 mL.
H. Trypsin Digestion
- Add Trypsin-TPCK (1 mg/mL stock in 1 mM HCl) at 1 mg: 37.5 mg (enzyme:substrate). This step requires 2.7 µL of 1 mg/mL Trypsin stock for 0.1 mg lysate. Digest overnight at room temperature with mixing.
- Continue through the C18 column purification, IAP, and C18 tip protocols before conducting the SECONDARY DIGESTION with trypsin. See the "Secondary Trypsin Digestion" section at the end of the protocol.
II. C18 Purification of Lysate Peptides
NOTE: Purification of peptides is performed at room temperature on C18 reversed-phase columns.
A. Solutions and Reagents
NOTE: Prepare all solutions with MS-grade or equivalent grade water (#27732), Trifluoroacetic Acid (TFA), and Acetonitrile (ACN). All percentage specifications are vol/vol. Prepare tubes containing organic solvents immediately before use and keep capped as much as possible to minimize evaporation.
- 20% TFA: Add 1 mL TFA to 4 mL water for a final volume of 5 mL.
- Solvent A (0.1% TFA): Add 0.25 mL of 20% TFA to 49.75 mL of water for a final volume of 50 mL.
- Solvent B (0.1% TFA, 40% ACN): Add 0.05 mL of 20% TFA to 6 mL of water and 4 mL of ACN for a final volume of 10 mL
- Wash Buffer (0.1% TFA, 5% ACN): Add 0.05 mL of 20% TFA to 9.5 mL of water and 0.5 mL of ACN for a final volume of 10 mL.
B. Acidification of Digested Cell Lysate
NOTE: Before loading the peptides from the digested sample on the column, they must be acidified with TFA for efficient peptide binding. The acidification step helps remove fatty acids from the digested peptide mixture.
- Add 1/20 volume of 20% TFA to the digest for a final concentration of 1% TFA. Check the pH by spotting a small amount of peptide sample on a pH strip (the pH should be < 3). After acidification, allow precipitate to form by letting sample stand for 15 min on ice.
- Centrifuge the acidified peptide solution at 4,000 - 10,000 x g for 15 min at room temperature to remove the precipitate. Transfer peptide-containing supernatant into a new microcentrifuge tube without dislodging the precipitated material.
C. Peptide Purification
NOTE: Application of all solutions, especially during the peptide loading and elution steps, should be performed slowly at approximately one drop per sec.
- Pre-wet the column with 0.5 mL 100% ACN.
- Equilibrate the column with 1 mL of Solvent A (0.1% TFA). Repeat this step once.
- Load acidified and cleared digest (from Section B)
- Wash with 1 mL of Solvent A (0.1% TFA). Repeat this step once.
- Wash with 0.5 mL of Wash Buffer (5% ACN, 0.1% TFA).
- Place columns above new 1.5 mL polypropylene tubes to collect eluate. Elute peptides with a sequential wash of 3 x 0.2 mL of Solvent B (0.1% TFA, 40% ACN).
- Dry the peptide solution in a vacuum concentrator set to ambient temperature overnight or until completely dry.
NOTE: Peptide solutions may be frozen at -80°C for one hr or longer before placing in the vacuum centrifuge; this step can prevent full tubes from spilling when placed at an angle to dry.
NOTE: A standard lyophilization apparatus is also acceptable in place of a vacuum concentrator.
NOTE: Dry, digested peptides are stable at -80°C for several months (seal the closed tube with parafilm for storage). The PTMScan® protocol can be interrupted before or after drying. Once the dried peptide is dissolved in IAP buffer (see next section), continue to the end of the procedure.
III. Immunoaffinity Purification (IAP)
A. Solutions and Reagents
NOTE: Prepare all solutions with MS-grade or equivalent grade water and TFA. All percentage specifications are vol/vol.
Materials Provided in the PTMScan® Kit PTMScan® IAP Buffer (10X) #9993 to 1X concentration. Store 1X buffer for up to one month at 4°C.
B. Procedure
- Centrifuge the tube containing lyophilized peptide at 2,000 x g for five min at room temperature to collect all material for dilution in IAP buffer. Add 1.4 mL IAP buffer. Resuspend pellets by pipetting repeatedly or vortexing until peptide is solubilized. Transfer solution to a 1.7 mL microcentrifuge tube.
- Clear solution by centrifugation at 10,000 x g for five min at 4°C in a microcentrifuge. The insoluble pellet may appear considerable. This will not pose a problem since most of the peptide will be soluble. Cool on ice.
- Centrifuge the vial of antibody-bead slurry at 2,000 x g for 30 sec and remove all buffer from the beads. Wash antibody beads four times with 1 mL of 1X PBS. Centrifuge at 2,000 x g after each wash. Resuspend beads in 40 µL PBS in the provided vial
- Transfer the peptide solution into the vial containing motif antibody beads. Pipette sample directly on top of the beads at the bottom of the tube to ensure immediate mixing. Avoid creating bubbles upon pipetting.
- Tighten the cap on the vial. Seal the top of the vial with parafilm to avoid leakage. Incubate on a rotator for two hr at 4°C.
- Centrifuge at 2,000 x g for 30 sec. If desired, save the supernatant for subsequent IAPs by storing it at -80°C.
- Add 1 mL of IAP buffer to the beads, mix by inverting the tube five times, centrifuge at 2,000 x g for 30 sec, and discard supernatant.
- Repeat Step 7 once for a total of TWO IAP buffer washes.
- Add 1 mL chilled HPLC water to the beads, mix by inverting the tube five times, centrifuge at 2,000 x g for 30 sec, and discard supernatant.
- Repeat Step 9 two times for a total of THREE water washes. After the last water wash, carefully remove all remaining supernatant before the next step.
- Add 55 µL of 0.15% TFA to the beads, tap the bottom of the tube several times (do not vortex), and let stand at room temperature for 10 min, mixing gently every 2-3 min.
- Centrifuge at 2,000 x g for 30 sec in a microcentrifuge and transfer supernatant to a new 1.7 mL microcentrifuge tube.
- Add 50 µL of 0.15% TFA to the beads, and repeat the centrifugation/elution step. Combine both eluents in the same 1.7 mL tube. Briefly centrifuge the eluent to pellet any remaining beads and carefully transfer eluent to a new 1.7 mL tube, taking care not to transfer any beads.
NOTE: After dissolving the peptide, check the pH of the peptide solution by spotting a small volume on pH indicator paper. The pH should be close to neutral, or no lower than 6.0. In the rare case that the pH is more acidic, titrate the peptide solution with 1 M Tris base solution that has not been adjusted for pH. Usually, 5-10 µL is sufficient to neutralize the solution.
NOTE: If 0.1 mg of peptide was initially purified, a fraction can be aliquoted into a separate tube for carbamidomethyl cysteine enrichment. For example, if the 0.1 mg peptides were initially resuspended in 1 mL IAP buffer for 0.1 mg/mL peptide concentration, transfer 0.1 mL of the 0.1 mg/mL clarified peptides into a new tube for 0.01 mg peptide amount and adjust the total volume to 1.4 mL. The remaining peptide can be frozen for later use.
NOTE: Perform all subsequent wash steps at 2-4°C. Avoid removing the last few microliters for all the washes except the final wash, since this may cause inadvertent bead removal.
NOTE: In this step, the post-translationally modified peptides of interest will be in the eluent.
IV. StageTip Purification and Secondary Trypsin Digestion of Peptides for LC-MS Analysis
NOTE: We recognize there are many other routine methods for concentrating peptides using commercial products, such as C18 tips (#41745), that have been optimized for peptide desalting/concentration. Regardless of the method used, we recommend that it be optimized for recovery and amenable to peptide loading capacities of at least 10 µg.
A. Solutions and Reagents
NOTE: Prepare all solutions with MS-grade or equivalent grade water (#27732), TFA, and ACN. Prepare tubes containing organic solvents immediately before use and keep capped as much as possible to minimize evaporation.
- Solvent C (0.1% TFA, 50% ACN): Add 10 µL of 100% TFA to 4 mL MS-grade water, then add 5 mL ACN, and adjust the final volume to 10 mL with MS-grade water.
- Solvent D (0.1% TFA): Add 10 µL of 100% TFA to 10 mL with MS-grade water.
- Solvent E (0.1% TFA, 40% ACN): Add 10 µL of 100% TFA to 3 mL MS-grade water, add 4 mL ACN, and adjust the final volume to 10 mL with MS-grade water.
- Equilibrate the C18 tip by passing 50 µL of Solvent C through once, followed by 50 µL of Solvent D two times.
- Load sample by passing IP eluent through the C18 tip. Load IAP eluent in two steps, using 50 µL in each step.
- Wash the C18 tip by passing 50 µL of Solvent D through twice.
- Elute peptides off the C18 tip by passing 10 µL of Solvent E through twice. Pool the resulting eluates.
- Dry down the C18 tip eluent in a vacuum concentrator.
- Thaw 1 M ammonium bicarbonate stock solution #78450.
- Prepare digestion buffer (50 mM ammonium bicarbonate, 5% ACN).
- Dilute PTMScan® Trypsin, TPCK-Treated #56296 with MS-grade water to make 0.5 µg/µL (e.g., add 40 µL water to 20 µg trypsin).
- Dilute the 0.5 µg/µL aliquot of trypsin with digestion buffer for a final concentration of 25 ng/µL (e.g., add 5 µL of 0.5 µg/µL trypsin to 95 µL digestion buffer).
- Resuspend the dried peptides from Step 5 with 10 µL of trypsin solution from Step 9. Vortex to redissolve the peptides and briefly centrifuge the sample to collect peptides/trypsin solution at the bottom of the microcentrifuge tube.
- Incubate the solution at 37°C for two hr.
- After trypsin digestion, add 1 µL of 5% TFA to the 10 µL digest solution. Vortex to mix and briefly centrifuge to collect peptide solution at the bottom of the microcentrifuge tube.
- Use a new C18 tip and repeat Step 1 to equilibrate with Solvent C, then wash twice with Solvent D.
- Transfer the 11 µL of acidified peptide solution to a newly conditioned C18 tip, rinse the microcentrifuge tube with 40 µL of Solvent D, and apply the rinse solution to the C18 tip for a final volume of 51 µL.
- Wash the C18 tip by passing 50 µL of Solvent D through twice.
- Elute peptides off the C18 tip by passing Solvent E through twice. Dry purified peptides under vacuum prior to LC-MS analysis.
B. Procedure
For Research Use Only. Not for Use in Diagnostic Procedures.