Cell Cycle Regulation
CST signaling pathway diagrams allow you to click on individual nodes to find research resources or product information. You can also download the pathway diagrams for educational and research purposes.
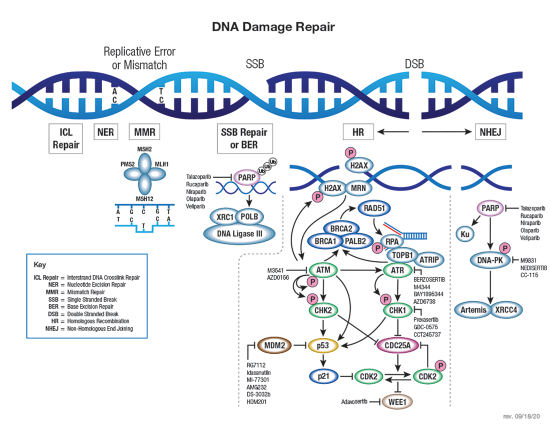
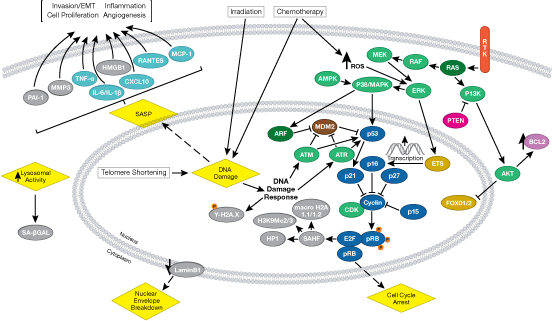
Control of eukaryotic cell growth and division involves molecular circuits known as “checkpoints” that ensure proper timing of cellular events. Passage through a checkpoint from one cell cycle phase to the next requires a coordinated set of proteins that monitor cell growth and DNA integrity. Uncontrolled cell division or propagation of damaged DNA can contribute to genomic instability and tumorigenesis.
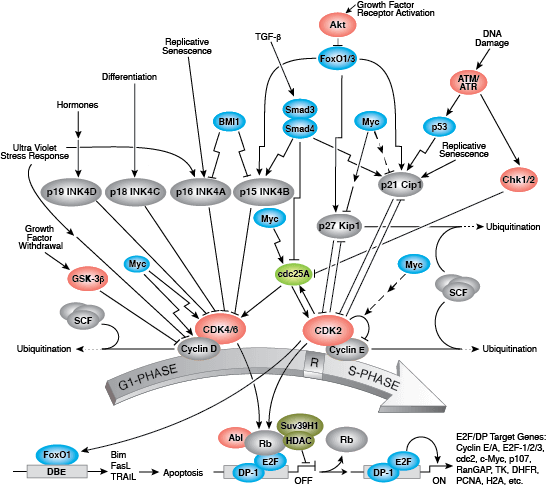
The G1/S checkpoint controls progression of cells through the restriction point (R) into the DNA synthesis S-phase. During G1, the tumor suppressor Rb binds and inhibits transcription factor E2F. Phosphorylation of Rb by cyclin-bound cyclin dependent kinases (CDK) in late G1 induces dissociation of Rb and permits E2F-mediated transcription of S-phase-promoting genes. Responding to upstream signals, INK4 and Kip/Cip family inhibitors control CDK activity and prevent entry into S-phase. DNA damage activates response pathways through ATM/ ATR and Chk1/2 kinases to block CDK activity, leading to cell cycle arrest and DNA repair or cell death.
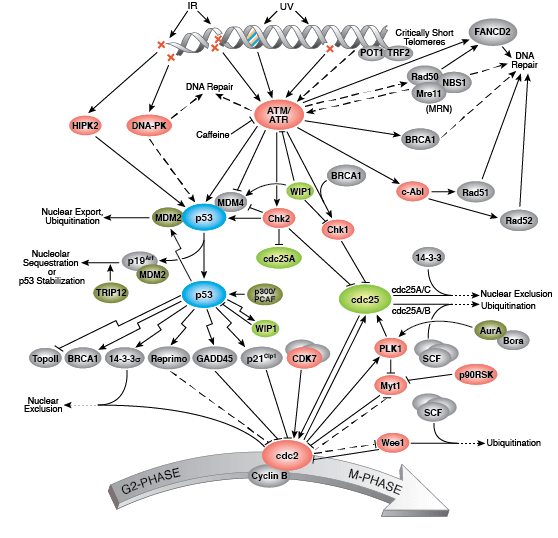
The G2/M checkpoint prevents cells containing damaged DNA from entering mitosis (M). Activated CDK1 (cdc2) bound to cyclin B promotes entry into M-phase. Wee1 and Myt1 kinases and cdc25 phosphatase competitively regulate CDK1 activity; Wee1 and Myt1 inhibit CDK1 and prevent entry into M-phase, while cdc25 removes inhibitory phosphates. DNA damage activates multiple kinases that phosphorylate kinases Chk1/2 and tumor suppressor protein p53. Chk1/2 kinases stimulate Wee1 activity and inhibit cdc25C, preventing entry into M-phase. Phosphorylation of p53 promotes dissociation between p53 and MDM2 and allows binding of the transcription factor to DNA.
The spindle checkpoint ensures proper chromatid attachment prior to progression from metaphase to anaphase. The SCF and APC/C protein complexes play prominent roles, with APC-cdc20 initiating the entry into anaphase by promoting ubiquitin-mediated degradation of multiple substrates, including cyclin B and the regulatory protein securin.
References:
- Cimprich KA, Cortez D (2008) ATR: an essential regulator of genome integrity. Nat. Rev. Mol. Cell Biol. 9(8), 616–27.
- van den Heuvel S, Dyson NJ (2008) Conserved functions of the pRB and E2F families. Nat. Rev. Mol. Cell Biol. 9(9), 713–24.
- Reinhardt HC, Yaffe MB (2009) Kinases that control the cell cycle in response to DNA damage: Chk1, Chk2, and MK2. Curr. Opin. Cell Biol. 21(2), 245–55.
- Malumbres M, Barbacid M (2009) Cell cycle, CDKs and cancer: a changing paradigm. Nat. Rev. Cancer 9(3), 153–66.
- Lindqvist A, RodrÃguez-Bravo V, Medema RH (2009) The decision to enter mitosis: feedback and redundancy in the mitotic entry network. J. Cell Biol. 185(2), 193–202.
- Wohlbold L, Fisher RP (2009) Behind the wheel and under the hood: functions of cyclin-dependent kinases in response to DNA damage. DNA Repair (Amst.) 8(9), 1018–24.
- Skaar JR, Pagano M (2009) Control of cell growth by the SCF and APC/C ubiquitin ligases. Curr. Opin. Cell Biol. 21(6), 816–24.
- Kim S, Yu H (2011) Mutual regulation between the spindle checkpoint and APC/C. Semin. Cell Dev. Biol. 22(6), 551–8.
- Musacchio A (2011) Spindle assembly checkpoint: the third decade. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 366(1584), 3595–604.
- Lord CJ, Ashworth A (2012) The DNA damage response and cancer therapy. Nature 481(7381), 287–94.