In order to derive the most information from samples related to neurodegenerative disease, it is important to consider:
Addressing and aligning these factors beforehand will allow the maximal amount of data to be obtained from each experiment.
A broad range of techniques are used to study neurodegenerative diseases including basic cellular and molecular protocols, electrophysiological assessments, and imaging-based methods that utilize antibody-based technology in situ.
Histological staining post-mortem is the most robust method used to identify proteins of interest and their subcellular localization and to visualize how these proteins interact.
Individual neuronal and glial populations can be isolated and characterized, along with their axons, dendrites, receptors, and neurotransmitter content. In addition, proteins that are altered in diseases states can be selectively marked.
Preparation of the tissues of the central nervous system require care to produce optimal results when staining. To make high quality neuropathological observations, an understanding of neuroanatomy and several simple, extra steps should be considered for studies in rodents: quick and complete exsanguination, careful removal of the brain, and rapid immersion in fixative. The spinal cord can be removed intact by using a syringe in a caudal to rostral manner.
Tissue samples are often prepared by quick freezing in optimal cutting temperature (OCT) medium (for use with IF) or by embedding in paraffin (for use with IHC). Section samples and allow to air dry on the slide before proceeding with the staining procedure.
Immunohistochemistry (IHC) can be used for anatomical and morphological screening by H&E staining. Any autofluorescence observed on an H&E stained slide under a FITC filter is indicative of degenerative neurons, while Fluoro Jade B stain is a more sensitive stain to detect neurodegeneration in the dendrites and axons. LFB-Holmes Silver stain can be utilized to visualize myelin and axons, while inflammation is visualized by staining for markers of astrocytes (GFAP) and microglia (CD68 and IBA1).
It is important to understand the application of a particular antibody - just because it performs as expected in a western blot, does not indicate a successful localization when performing IF staining. Therefore, it is important to verify application-specific validation. Additionally, it is important to choose primary-secondary pairs and fluorophore conjugates compatible with the imaging system.
Using rabbit monoclonal antibodies especially for IHC of mouse or human tissues has many advantages. For example, they provide increased affinity and specificity, decrease background, and are less likely to cross-react with antibodies from other species.
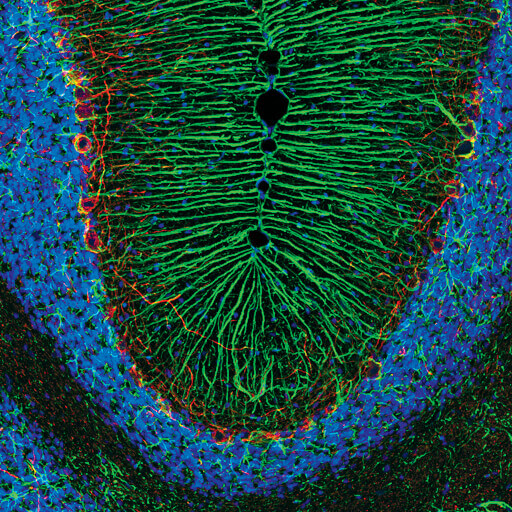
Confocal immunofluorescent analysis of rat cerebellum using GFAP (D1F4Q) XP® Rabbit mAb (green) and Neurofilament-H (RMdO 20) Mouse mAb #2836 (red). Blue pseudocolor = DRAQ5 #4084 (fluorescent DNA dye).
Primary cells isolated from freshly harvested brain or spinal cord, or from cerebrospinal fluid, as well as induced pluripotent stem cells (iPSCs) can be prepared for ICC analysis. It is important to use coated slides, fix the cells, permeabilize, block, and then stain. For multiplexing, special consideration should be given to the wavelengths of excitation sources and spectral characteristics of filter sets in order to minimize spectral overlap.
Flow cytometry has been used to perform analysis of microparticles and lipoprotein particles associated with neurodegenerative diseases including ApoE4 and complexes with Aβ. In addition, deep immunophenotyping of cell types in neurological disorders and analysis of pro- and anti-inflammatory cytokine production can be assessed by flow cytometric analysis.
The complexity of the affected proteins, as well as the sometimes barely detectable circulating levels has resulted in the use of ELISA to predominantly measure neurotransmitter output as well as for signs of neuroinflammation by examining pro- and anti-inflammatory cytokine levels.