BAR Protein Domain

Amphiphysin BAR Domain From Drosophila.
Domain Binding and Function
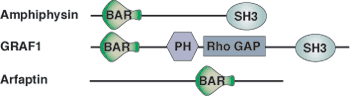
The BAR (Bin/Amphiphysin/Rvs) domain is found in over 35 proteins encoded in the human genome. These proteins function in diverse cellular processes such as endocytosis (i.e. endophilins, sorting nexins, and amphiphysin) and actin reorganization (i.e. RhoGAPs and RhoGEFs). The BAR domain of Arfaptin has been shown to bind Rac1. The functional BAR dimer is a banana shaped bundle of six helices with positive charges on the tips and along its concave surface that may mediate phospholipid binding. The curvature of the concave surface would fit a rounded membrane with a diameter of ~ 220 angstroms. While all BAR domains probably bind curved lipids, a subset of BAR domains can actually induce membrane curvature. It is therefore predicted that BAR domains may function during vesiculation to either target proteins to curved membranes or to physically assist in inducing membrane curvature.
Structure
The BAR domain of amphiphysin is approximately 210 amino acids in length and consists of a coiled coil composed of three extended α helices. Two monomers dimerize to form the functional banana shaped bundle of six helices. Positive charges are clustered at the tips of the banana like structure and along its concave surface. These positive charges are thought to mediate binding to phospholipids where the curvature of the concave surface would fit a rounded membrane with a diameter of ~ 220 angstroms.
Structure Reference
- Peter, B.J. et al. (2004) Science 303(5657), 495–499.
Examples of Domain Proteins