GPCR, Calcium, cAMP
CST signaling pathway diagrams allow you to click on individual nodes to find research resources or product information. You can also download the pathway diagrams for educational and research purposes.
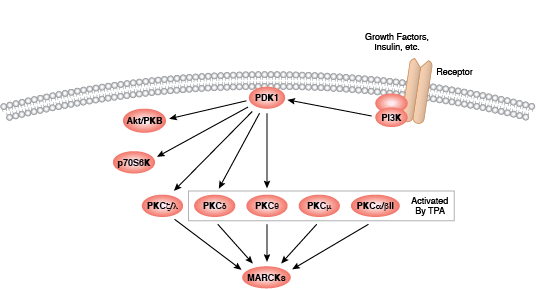
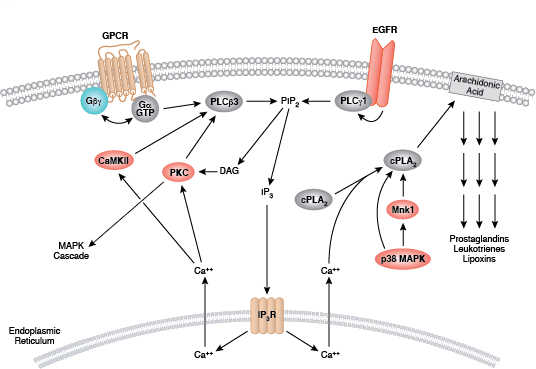
Protein kinase C (PKC) family members regulate numerous cellular responses including secretion, gene expression, cell proliferation, and the inflammation response. Basic protein structure includes an amino-terminal regulatory region connected to a carboxy-terminal kinase domain by a hinge region. PKC enzymes contain an auto-inhibitory pseudosubstrate domain that binds a catalytic domain sequence to inhibit kinase activity. Binding of second messenger activators localizes PKC enzymes to the cell membrane where allosteric changes allow pseudosubstrate release from the catalytic sequence. Differences among PKC regulatory regions allow for variable second messenger binding and are the basis for the division of the PKC family into 3 broad groups. Conventional PKC enzymes (cPKC; isoforms PKCα, PKCβ, and PKCγ) contain functional C1 and C2 regulatory domains; cPKC enzyme activation requires binding of diacylglycerol (DAG) and a phospholipid to the C1 domain, and Ca2+ binding to the C2 domain. Novel PKC enzymes (nPKC; isoforms PKCδ, PKCε, PKCη, and PKCθ) also require DAG binding for activation but contain a novel C2 domain that does not act as a Ca2+ sensor. Distantly related protein kinase D proteins are often associated with novel PKC enzymes as they respond to DAG but not Ca2+ stimulation. Atypical enzymes (aPKC; isoforms PKCζ and PKCι/λ) contain a non-functional C1 domain and lack a C2 domain, requiring no second messenger binding for aPKC activation.
Activation of PKC enzymes requires a series of phosphorylation events, association with additional proteins, and allosteric changes. All isoforms require phosphorylation at conserved sites within the catalytic domain active loop and turn motifs; conventional and novel enzymes require the additional phosphorylation of a site within a catalytic region hydrophobic motif. Following translation, nascent PKC localizes to the cell membrane where it associates with Hsp90 and Cdc37. Binding of these co-chaperones to the kinase domain precedes phosphorylation of the active loop site by PDK1. Second messengers activate PKC by recruiting the cytosolic enzyme to the plasma membrane where allosteric changes release the pseudosubstrate from the catalytic domain sequence.
Additional phosphorylation events regulate PKC activity by controlling protein interaction, stability, and localization. For example, stimulus-specific phosphorylation of PKCδ can result in PKCδ degradation, cleavage, or increased activity. In response to DNA damage and oxidative stress, PKCδ promotes apoptosis through activation of the p53-mediated pathway and inhibition of prosurvival proteins such as Akt, Cdk1, and cyclin D1. Transduction through the TNF pathway promotes a pro-survival response as PKCδ activates the Akt, NF-κB, and MEK pathways.
References:
- Newton AC (2010) Protein kinase C: poised to signal. Am. J. Physiol. Endocrinol. Metab. 298(3), E395–402.
- Newton PM, Messing RO (2010) The substrates and binding partners of protein kinase Cepsilon. Biochem. J. 427(2), 189–96.
- Freeley M, Kelleher D, Long A (2011) Regulation of Protein Kinase C function by phosphorylation on conserved and non-conserved sites. Cell. Signal. 23(5), 753–62.
- Basu A, Pal D (2010) Two faces of protein kinase Cδ: the contrasting roles of PKCδ in cell survival and cell death. ScientificWorldJournal 10, 2272–84.
- Rozengurt E (2011) Protein kinase D signaling: multiple biological functions in health and disease. Physiology (Bethesda) 26(1), 23–33.