| Cat. # | Size | Qty. | Price |
|---|---|---|---|
| 9095S | 1 Kit (1000 assays (96 well format)) |
|
| Product Includes | Quantity (with Count) | Solution Color | |||
|---|---|---|---|---|---|
| XTT Reagent | 2 x 25 ml | ||||
| Electron Coupling Solution | 2 x 0.5 ml | Yellow |
Product Information
NOTE: Precipitation may occur when reagents are stored at −20°C. Make sure reagents are clear prior to use. If necessary, warm reagents to 37°C to reconstitute.
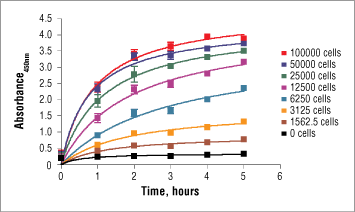
NOTE: The optimal incubation time for this assay depends on experimental setup, such as: cell type, cell number, and treatment. Optimization of incubation time can be determined by reading one plate at various time points after addition of XTT detection solution (featured below).
posted October 2012
Protocol Id: 46
All Species Expected
Cell viability and proliferation assays are widely used in drug discovery for the study of growth factors, cytokines, and cytotoxic agents. High throughput screening, in early drug discovery compound screening and in later drug safety and toxicity studies, requires a reliable, sensitive, and simple assay with the ability to analyze a large number of samples. Colorimetric cell viability assays using tetrazolium salt, such as MTT, XTT, WST-1, etc. were developed based on live cells reduction of tetrazolium salt into highly colored formazan compounds (1,2). In contrast to cell proliferation assays, such as radioactive thymidine or BrdU labeling of DNA in live cells followed by quantification of the incorporated thymidine (by quantifying sample radioactivity) or BrdU (using anti-BrdU antibody), the XTT assay doesn’t require radioactive materials, cell fixing, or cell permeabilization. Thus, unlike alternative cellular analysis assays, cells examined in the XTT assay may be used for further analysis.
Except as otherwise expressly agreed in a writing signed by a legally authorized representative of CST, the following terms apply to Products provided by CST, its affiliates or its distributors. Any Customer's terms and conditions that are in addition to, or different from, those contained herein, unless separately accepted in writing by a legally authorized representative of CST, are rejected and are of no force or effect.
Products are labeled with For Research Use Only or a similar labeling statement and have not been approved, cleared, or licensed by the FDA or other regulatory foreign or domestic entity, for any purpose. Customer shall not use any Product for any diagnostic or therapeutic purpose, or otherwise in any manner that conflicts with its labeling statement. Products sold or licensed by CST are provided for Customer as the end-user and solely for research and development uses. Any use of Product for diagnostic, prophylactic or therapeutic purposes, or any purchase of Product for resale (alone or as a component) or other commercial purpose, requires a separate license from CST. Customer shall (a) not sell, license, loan, donate or otherwise transfer or make available any Product to any third party, whether alone or in combination with other materials, or use the Products to manufacture any commercial products, (b) not copy, modify, reverse engineer, decompile, disassemble or otherwise attempt to discover the underlying structure or technology of the Products, or use the Products for the purpose of developing any products or services that would compete with CST products or services, (c) not alter or remove from the Products any trademarks, trade names, logos, patent or copyright notices or markings, (d) use the Products solely in accordance with CST Product Terms of Sale and any applicable documentation, and (e) comply with any license, terms of service or similar agreement with respect to any third party products or services used by Customer in connection with the Products.